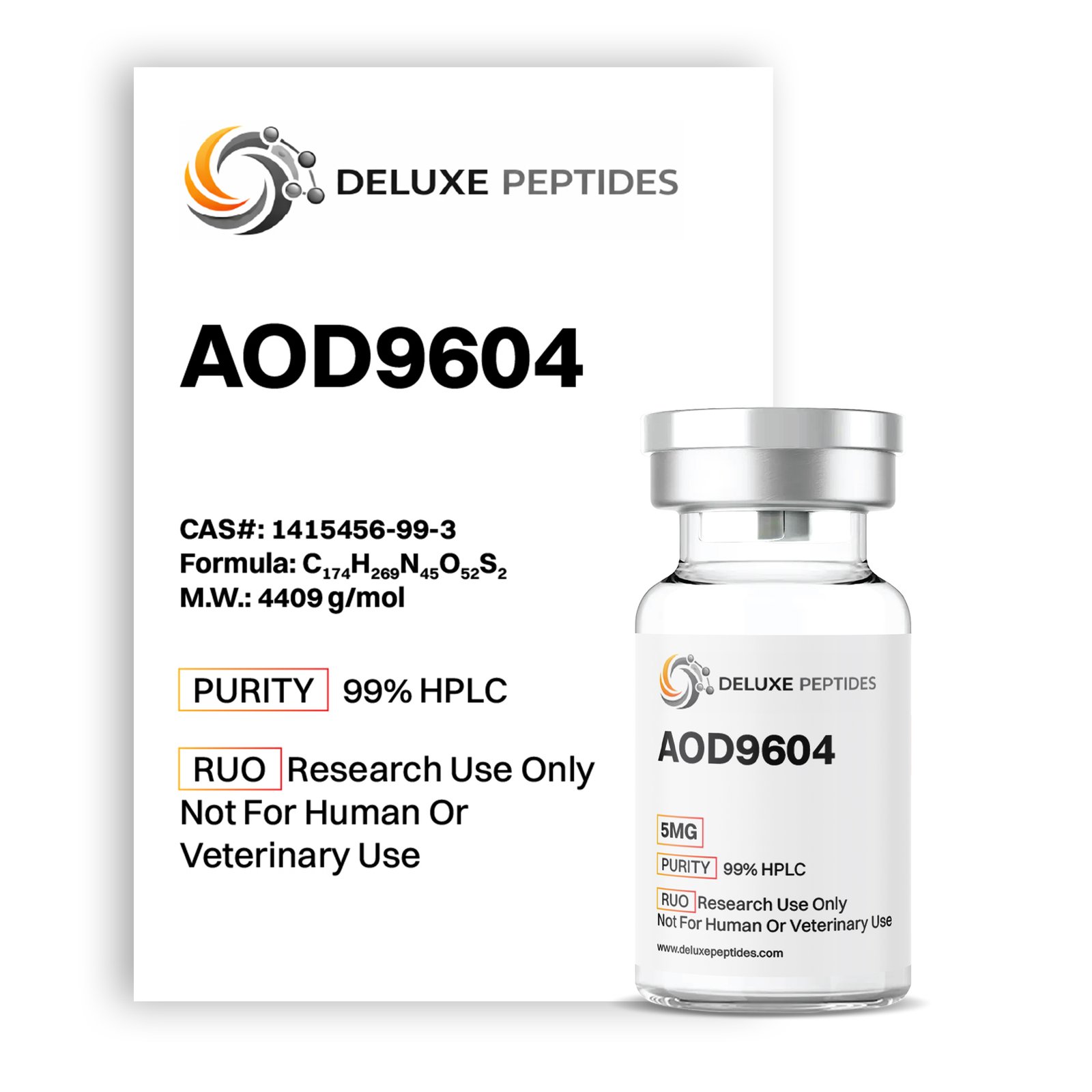
AOD9604 5mg – Research Overview
AOD9604 5mg is a synthetic peptide corresponding to amino acids 176–191 of the human growth hormone (hGH) sequence. In laboratory research, AOD9604 5mg is studied for its involvement in metabolic pathway modeling and adipose tissue signaling investigations. Unlike full-length growth hormone, AOD9604 represents a specific fragment designed to isolate and examine targeted signaling mechanisms in controlled experimental systems.
AOD9604 5mg is supplied in lyophilized form to preserve structural stability and ensure reliable peptide integrity prior to reconstitution. This compound is intended strictly for laboratory research and analytical applications.
Molecular Characteristics of AOD9604 5mg
AOD9604 is a modified fragment of the C-terminal region of human growth hormone, specifically amino acids 176–191. This region has been studied in research settings for its role in metabolic signaling distinct from the broader endocrine actions of intact growth hormone.
In laboratory environments, AOD9604 5mg is referenced in studies involving:
-
Adipocyte signaling pathway modeling
-
Lipid metabolism research
-
Growth hormone fragment receptor interaction
-
cAMP pathway modulation
-
Metabolic cascade investigation
Because AOD9604 is a fragment rather than a full-length hormone analog, it allows researchers to examine selective molecular interactions within metabolic research frameworks.
Mechanism of Action (Research Context)
In controlled in vitro models, AOD9604 5mg is studied for its interaction with metabolic signaling pathways related to adipose tissue regulation. Research investigations suggest that this fragment may influence intracellular cascades distinct from those activated by full-length growth hormone.
Experimental models have examined potential involvement in:
-
cAMP-mediated signaling pathways
-
Hormone-sensitive lipase pathway modeling
-
Adipocyte receptor interaction studies
-
Downstream transcriptional regulation
-
Metabolic gene expression frameworks
Importantly, AOD9604 5mg is studied in laboratory systems specifically to isolate fragment-mediated signaling effects separate from systemic endocrine modulation observed with intact growth hormone.
Researchers often compare fragment-based signaling to full-length hGH models to evaluate structural-function relationships within peptide hormone biology.
Research Applications
AOD9604 5mg is commonly referenced in laboratory research involving:
-
Adipose tissue signaling analysis
-
Growth hormone fragment pathway modeling
-
Metabolic cascade investigations
-
Lipid metabolism research frameworks
-
Intracellular second messenger pathway studies
-
Comparative peptide fragment analysis
In molecular biology laboratories, AOD9604 5mg serves as a tool compound for evaluating how specific hGH fragments may interact with metabolic signaling networks under structured experimental conditions.
For broader scientific context on growth hormone fragment research and metabolic pathway studies, researchers frequently consult peer-reviewed resources such as the National Center for Biotechnology Information (NCBI).
Distinction from Full-Length Growth Hormone
AOD9604 5mg differs structurally from intact human growth hormone. By isolating amino acids 176–191, this fragment allows researchers to study specific molecular interactions independent of the full endocrine profile associated with hGH receptor activation.
Laboratory comparisons may focus on:
-
Receptor binding selectivity
-
Intracellular cascade specificity
-
Metabolic pathway activation intensity
-
Gene transcription patterns
-
Structural-functional peptide relationships
This fragment-based approach is valuable for dissecting complex hormone signaling systems into more defined experimental components.
Stability and Laboratory Handling
AOD9604 5mg is supplied as a lyophilized sterile powder to enhance storage stability and reduce hydrolytic degradation prior to reconstitution.
For laboratory research use:
-
Store under recommended refrigerated conditions
-
Avoid repeated freeze–thaw cycles
-
Reconstitute using sterile laboratory-grade solvent
-
Follow institutional peptide handling protocols
Lyophilization supports long-term peptide stability and ensures reproducibility in structured experimental systems.
Proper storage and handling are essential to maintain peptide integrity and support consistent laboratory results.
Product Specifications
-
Compound: AOD9604
-
Sequence: hGH Fragment 176–191
-
Classification: Synthetic growth hormone fragment
-
Strength: 5mg
-
Form: Lyophilized sterile powder
-
Appearance: White to off-white solid
-
Storage: Refrigerated conditions recommended
-
Intended Use: Laboratory research only
AOD9604 5mg is formulated exclusively for professional research environments and scientific investigation.
Analytical Verification and Quality Standards
Each batch of AOD9604 5mg undergoes analytical verification to confirm identity and purity using validated laboratory methodologies. These procedures may include:
-
High-performance liquid chromatography (HPLC)
-
Mass spectrometry analysis
-
Structural sequence verification
-
Batch consistency validation
Lot identification numbers are assigned to ensure traceability and documentation within formal research protocols.
Quality control measures are implemented to maintain consistent production standards across batches.
Broader Scientific Context
AOD9604 5mg is widely referenced in laboratory research exploring metabolic signaling and peptide fragment functionality. By isolating a specific region of the human growth hormone sequence, researchers can evaluate targeted molecular interactions within controlled experimental frameworks.
Such studies contribute to a deeper understanding of peptide structure-function relationships and metabolic pathway regulation in vitro.
Compliance Notice
AOD9604 5mg is intended strictly for laboratory research use.
This product is not approved for human consumption, therapeutic application, or clinical use. AOD9604 5mg is sold exclusively for in vitro research, analytical testing, and scientific investigation by qualified professionals.


Reviews
There are no reviews yet.